Indian Plant Antiviral Compound Database (IMPAC)
| Indian Plant Antiviral Compound | Description |
|---|---|
| InAVPCDB Accession Number | AVD030 |
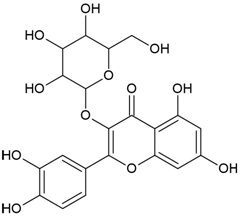
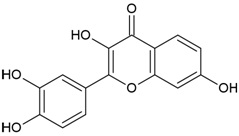
| Compound Name | Hyperoside |
| Phytochemical category | Flavanoid |
| PubChem Id | 5281643 |
| IUPAC Name | 2-(3,4-dihydroxyphenyl)-5,7-dihydroxy-3-[(2S,3R,4S,5R,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxychromen-4-one |
| Molecular Formula | C21H20O12 |
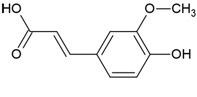
| Canonical SMILES Notation | C1=CC(=C(C=C1C2=C(C(=O)C3=C(C=C(C=C3O2)O)O)OC4C(C(C(C(O4)CO)O)O)O)O)O |
| Binomial Name | Azadirachta indica |
| Vernacular Names | Vepilai/Vepaku/Margosa/Neem |
| Plant Part | Leaf |
| Family | Meliaceae |
| Description | Influenza viruses and SARS-CoV-2. |
| Mechanism of action | It could antagonize N protein-induced S-phase arrest by interfering with interaction between N protein and p53 and inhibit viral replication. |
| Molecular weight | 464.38 g/mol |
| XLOGP3 | 0.36 |
| Num. H-bond acceptors | 12 |
| Num. H-bond donors | 8 |
| Rotatable bonds | 4 |
| Topological polar surface area | 210.51 Ų |
| Bioavailability Score | 0.17 |
| Druglikeness | No |
| Lipinski | 2 violations |
| References | Su M, Shi D, Xing X, Qi S, Yang D, Zhang J, Han Y, Zhu Q, Sun H, Wang X, Wu H. Coronavirus PEDV nucleocapsid protein interacts with p53 to induce cell cycle arrest in S-phase and promotes viral replication. Journal of Virology. 2021 May 26:JVI-00187. Halder P, Pal U, Paladhi P, Dutta S, Paul P, Pal S, Das D, Ganguly A, Ray A, Ghosh S. Evaluation of potency of the selected bioactive molecules from Indian medicinal plants with MPro of SARS-CoV-2 through in silico analysis. Journal of Ayurveda and integrative medicine. 2021 May 21. |