Indian Plant Antiviral Compound Database (IMPAC)
| Indian Plant Antiviral Compound | Description |
|---|---|
| InAVPCDB Accession Number | AVD038 |
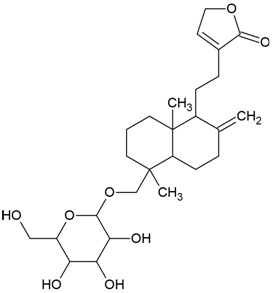
| Compound Name | Neoandrographolide |
| Phytochemical category | Diterpenoid |
| PubChem Id | 9848024 |
| IUPAC Name | 4-[2-[(1R,4aS,5R,8aS)-5,8a-dimethyl-2-methylidene-5-[[(2R,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxymethyl]-3,4,4a,6,7,8-hexahydro-1H-naphthalen-1-yl]ethyl]-2H-furan-5-one |
| Molecular Formula | C26H40O8 |
| Canonical SMILES Notation | CC1(CCCC2(C1CCC(=C)C2CCC3=CCOC3=O)C)COC4C(C(C(C(O4)CO)O)O)O |
| Binomial Name | Andrographis paniculata |
| Vernacular Names | Nilavembu/Hara chirayata/green chiretta/king of bitters |
| Plant Part | Leaf |
| Family | Acanthaceae |
| Description | Human immunodeficiency virus, Influenza A, Hepatitis B virus, Hepatitis C virus, Human papilloma virus, Herpes simplex viruses, and SARS-CoV-2. |
| Mechanism of action | It inhibits viral proteases and interrupts viral replication, fusion and adsorption of virus to the host cell, and binding to viral receptor and co-receptor; It inhibits enzymes involved in DNA/RNA/Genome replication by the virus, translation, post-translation and reverse transcription. |
| Molecular weight | 480.59 g/mol |
| XLOGP3 | 2.63 |
| Num. H-bond acceptors | 8 |
| Num. H-bond donors | 4 |
| Rotatable bonds | 7 |
| Topological polar surface area | 125.68 Ų |
| Bioavailability Score | 0.55 |
| Druglikeness | No |
| Lipinski | 0 violation |
| References | Jadhav AK, Karuppayil SM. Andrographis paniculata (Burm. F) Wall ex Nees: Antiviral properties. Phytotherapy Research. 2021 Apr 30. Computational investigation on Andrographis paniculata phytochemicals to evaluate their potency against SARS-CoV-2 in comparison to known antiviral compounds in drug trials. |