Indian Plant Antiviral Compound Database (IMPAC)
| Indian Plant Antiviral Compound | Description |
|---|---|
| InAVPCDB Accession Number | AVD018 |
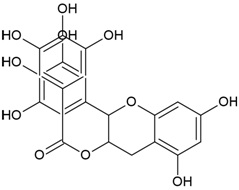
| Compound Name | Epigallocatechin gallate |
| Phytochemical category | Polyphenol |
| PubChem Id | 65064 |
| IUPAC Name | [(2R,3R)-5,7-dihydroxy-2-(3,4,5-trihydroxyphenyl)-3,4-dihydro-2H-chromen-3-yl] 3,4,5-trihydroxybenzoate |
| Molecular Formula | C22H18O11 |
| Canonical SMILES Notation | C1C(C(OC2=CC(=CC(=C21)O)O)C3=CC(=C(C(=C3)O)O)O)OC(=O)C4=CC(=C(C(=C4)O)O)O |
| Binomial Name | Camellia sinensis var. assamica |
| Vernacular Names | Teyilai/Chai/Black tea |
| Plant Part | Leaf |
| Family | Theaceae |
| Description | Influenza, Human immunodeficiency virus, Hepatitis C virus, and SARS-CoV-2. |
| Mechanism of action | Its viral efficacy of EGCG is attributable to damage to the physical properties of the viral envelope and partial inhibition of the Neuraminidase surface glycoprotein; and inhibits reverse transcriptase mechanism. |
| Molecular weight | 458.37 g/mol |
| XLOGP3 | 1.17 |
| Num. H-bond acceptors | 11 |
| Num. H-bond donors | 8 |
| Rotatable bonds | 4 |
| Topological polar surface area | 197.37 Ų |
| Bioavailability Score | 0.17 |
| Druglikeness | No |
| Lipinski | 2 violations |
| References | Kim M, Kim SY, Lee HW, Shin JS, Kim P, Jung YS, Jeong HS, Hyun JK, Lee CK. Inhibition of influenza virus internalization by (?)-epigallocatechin-3-gallate. Antiviral research. 2013 Nov 1;100(2):460-72. Zhong Y, Ma CM, Shahidi F. Antioxidant and antiviral activities of lipophilic epigallocatechin gallate (EGCG) derivatives. Journal of Functional Foods. 2012 Jan 1;4(1):87-93. Li S, Hattori T, Kodama EN. Epigallocatechin gallate inhibits the HIV reverse transcription step. Antiviral Chemistry and Chemotherapy. 2011 Aug;21(6):239-43. |