Indian Plant Antiviral Compound Database (IMPAC)
| Indian Plant Antiviral Compound | Description |
|---|---|
| InAVPCDB Accession Number | AVD010 |
| Compound Name | Chebulagic acid |
| Phytochemical category | Polyphenolic tannin |
| PubChem Id | 442674 |
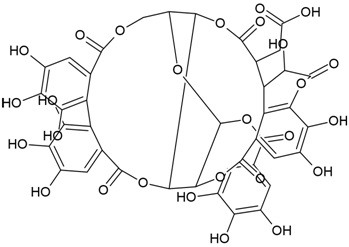
| IUPAC Name | 2-[(4R,5S,7R,25S,26R,29S,30S,31S)-13,14,15,18,19,20,31,35,36-nonahydroxy-2,10,23,28,32-pentaoxo-5-(3,4,5-trihydroxybenzoyl)oxy-3,6,9,24,27,33-hexaoxaheptacyclo[28.7.1.04,25.07,26.011,16.017,22.034,38]octatriaconta-1(37),11,13,15,17,19,21,34(38),35-nonaen-29-yl]acetic acid |
| Molecular Formula | C41H30O27 |
| Canonical SMILES Notation | C1C2C3C(C(C(O2)OC(=O)C4=CC(=C(C(=C4)O)O)O)OC(=O)C5=CC(=C(C6=C5C(C(C(=O)O3)CC(=O)O)C(C(=O)O6)O)O)O)OC(=O)C7=CC(=C(C(=C7C8=C(C(=C(C=C8C(=O)O1)O)O)O)O)O)O |
| Binomial Name | Terminalia chebula |
| Vernacular Names | Kadukkai/Karakkaya/Harad/Myrobalan |
| Plant Part | Fruit |
| Family | Combretaceae |
| Description | Human enterovirus 71, Herpes simplex virus type 1, Influenza viruses, and SARS-CoV-2. |
| Mechanism of action | It exert its inhibitory effect on neuraminidase-mediated viral release; by inhibting viral proteases. |
| Molecular weight | 954.66 g/mol |
| XLOGP3 | 0.36 |
| Num. H-bond acceptors | 27 |
| Num. H-bond donors | 13 |
| Rotatable bonds | 5 |
| Topological polar surface area | 447.09 Ų |
| Bioavailability Score | 0.11 |
| Druglikeness | No |
| Lipinski | 3 violations |
| References | Li P, Du R, Wang Y, Hou X, Wang L, Zhao X, Zhan P, Liu X, Rong L, Cui Q. Identification of chebulinic acid and chebulagic acid as novel influenza viral neuraminidase inhibitors. Frontiers in microbiology. 2020 Feb 28;11:182. Du R, Cooper L, Chen Z, Lee H, Rong L, Cui Q. Discovery of chebulagic acid and punicalagin as novel allosteric inhibitors of SARS-CoV-2 3CLpro. Antiviral research. 2021 Jun 1;190:105075. |