| Indian Plant Antiviral Compound |
Description |
| InAVPCDB Accession Number | AVD013 |
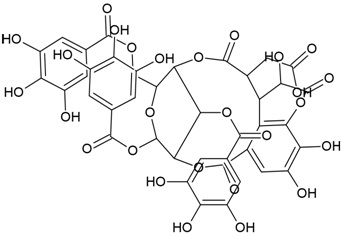
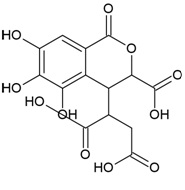
| Compound Name | Corilagin |
| Phytochemical category | Flavanoid |
| PubChem Id | 73568 |
| IUPAC Name | [(1S,19R,21S,22R,23R)-6,7,8,11,12,13,22,23-octahydroxy-3,16-dioxo-2,17,20-trioxatetracyclo[17.3.1.04,9.010,15]tricosa-4,6,8,10,12,14-hexaen-21-yl] 3,4,5-trihydroxybenzoate |
| Molecular Formula | C27H22O18 |
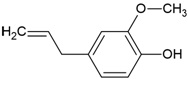
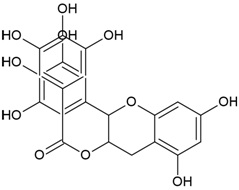
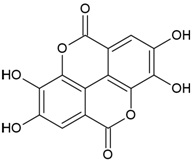
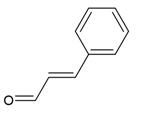
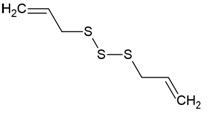
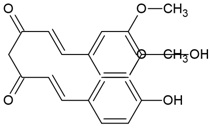
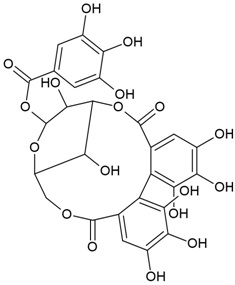
| Canonical SMILES Notation | C1C2C(C(C(C(O2)OC(=O)C3=CC(=C(C(=C3)O)O)O)O)OC(=O)C4=CC(=C(C(=C4C5=C(C(=C(C=C5C(=O)O1)O)O)O)O)O)O)O |
| Binomial Name | Phyllanthus amarus |
| Vernacular Names | Kizhanelli/Nela Usiri/Bhui Amala |
| Plant Part | Leaf |
| Family | Phyllanthaceae |
| Description | Hepatitis viruses, Human enterovirus, Coxsackievirus and SARS-CoV-2 |
| Mechanism of action | It inhibits viral proteases and reverse trancriptases; restricts viral replicaiton. |
| Molecular weight | 634.45 g/mol |
| XLOGP3 | 0.07 |
| Num. H-bond acceptors | 18 |
| Num. H-bond donors | 11 |
| Rotatable bonds | 3 |
| Topological polar surface area | 310.66 Ų |
| Bioavailability Score | 0.17 |
| Druglikeness | No |
| Lipinski | 3 violations |
| References | Yeo SG, Song JH, Hong EH, Lee BR, Kwon YS, Chang SY, Kim SH, won Lee S, Park JH, Ko HJ. Antiviral effects of Phyllanthus urinaria containing corilagin against human enterovirus 71 and Coxsackievirus A16 in vitro. Archives of pharmacal research. 2015 Feb;38(2):193-202. Hiremath S, Kumar HV, Nandan M, Mantesh M, Shankarappa KS, Venkataravanappa V, Basha CJ, Reddy CL. In silico docking analysis revealed the potential of phytochemicals present in Phyllanthus amarus and Andrographis paniculata, used in Ayurveda medicine in inhibiting SARS-CoV-2. 3 Biotech. 2021 Feb;11(2):1-8. |