| Indian Plant Antiviral Compound |
Description |
| InAVPCDB Accession Number | AVD032 |
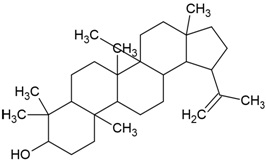
| Compound Name | Lupeol |
| Phytochemical category | Triterpenoid |
| PubChem Id | 259846 |
| IUPAC Name | (1R,3aR,5aR,5bR,7aR,9S,11aR,11bR,13aR,13bR)-3a,5a,5b,8,8,11a-hexamethyl-1-prop-1-en-2-yl-1,2,3,4,5,6,7,7a,9,10,11,11b,12,13,13a,13b-hexadecahydrocyclopenta[a]chrysen-9-ol |
| Molecular Formula | C30H50O |
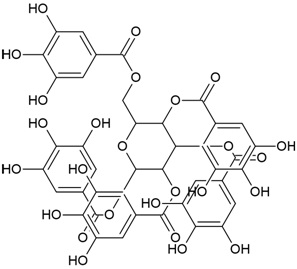
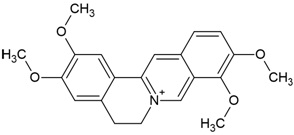
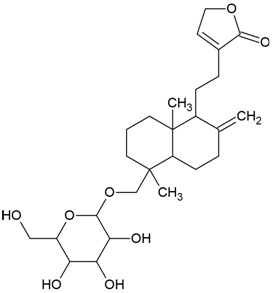
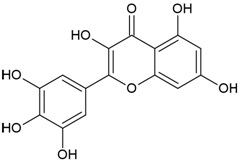
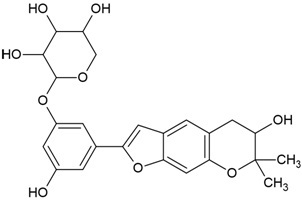
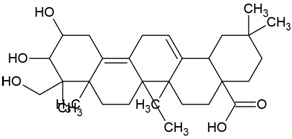
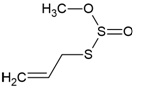
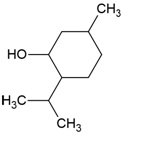
| Canonical SMILES Notation | CC(=C)C1CCC2(C1C3CCC4C5(CCC(C(C5CCC4(C3(CC2)C)C)(C)C)O)C)C |
| Binomial Name | Decalepis hamiltonii |
| Vernacular Names | Mahali kizhangu/Nannari kommulu/Mahannikizhangu |
| Plant Part | Tuber |
| Family | Apocynaceae |
| Description | Dengue virus, Herpes virus, Ranikhet disease, Encephalomyocarditis, Semiliki Forest virus, and SARS-CoV-2 virus. |
| Mechanism of action | It works by modulating the expression or activity of several molecules such as cytokines IL-2, IL4, IL5, IL?, proteases, ?-glucosidase, cFLIP, Bcl-2 and NF?B |
| Molecular weight | 426.72 g/mol |
| XLOGP3 | 9.87 |
| Num. H-bond acceptors | 1 |
| Num. H-bond donors | 1 |
| Rotatable bonds | 1 |
| Topological polar surface area | 20.23 Ų |
| Bioavailability Score | 0.55 |
| Druglikeness | No |
| Lipinski | 1 violation |
| References | Silva FC, Rodrigues VG, Duarte LP, Lula IS, Sinisterra RD, Vieira-Filho SA, Rodrigues RA, Kroon EG, Oliveira PL, Farias LM, Magalhaes PP. Antidiarrheal activity of extracts from Maytenus gonoclada and inhibition of Dengue virus by lupeol. Anais da Academia Brasileira de Ci?ncias. 2017 Jul;89:1555-64. Tolo FM, Rukunga GW, Muli FW, Ochora JM, Irungu BN, Muthaura CN, Wanjiku CK, Mungai GM, Ngoc Q, Hashimoto K, Asakawa Y. The antiviral activity of compounds isolated from Kenyan Carissa edulis (Forssk.) Vahl. Journal of Medicinal Plants Research. 2010 Aug 4;4(15):1517-22. |